International Monitoring Expands After Cruise Ship Hantavirus Cases
Health authorities worldwide are monitoring passengers and tracing contacts after a deadly Andes hantavirus outbreak aboard the M/V Hondius cruise ship.
![]() May 12
May 12
by Reid Ashton
Home > Health

Health authorities worldwide are monitoring passengers and tracing contacts after a deadly Andes hantavirus outbreak aboard the M/V Hondius cruise ship.
![]() May 12
May 12
by Reid Ashton

CDC halts testing for infectious diseases due to staffing shortages, raising concerns over U.S. public health surveillance and outbreak response in 2026
![]() April 6
April 6
by Reid Ashton

A family raising twins with profound autism shares their daily challenges as experts and advocates debate creating a separate diagnosis for severe autism.
![]() Feb 17
Feb 17
by Reid Ashton
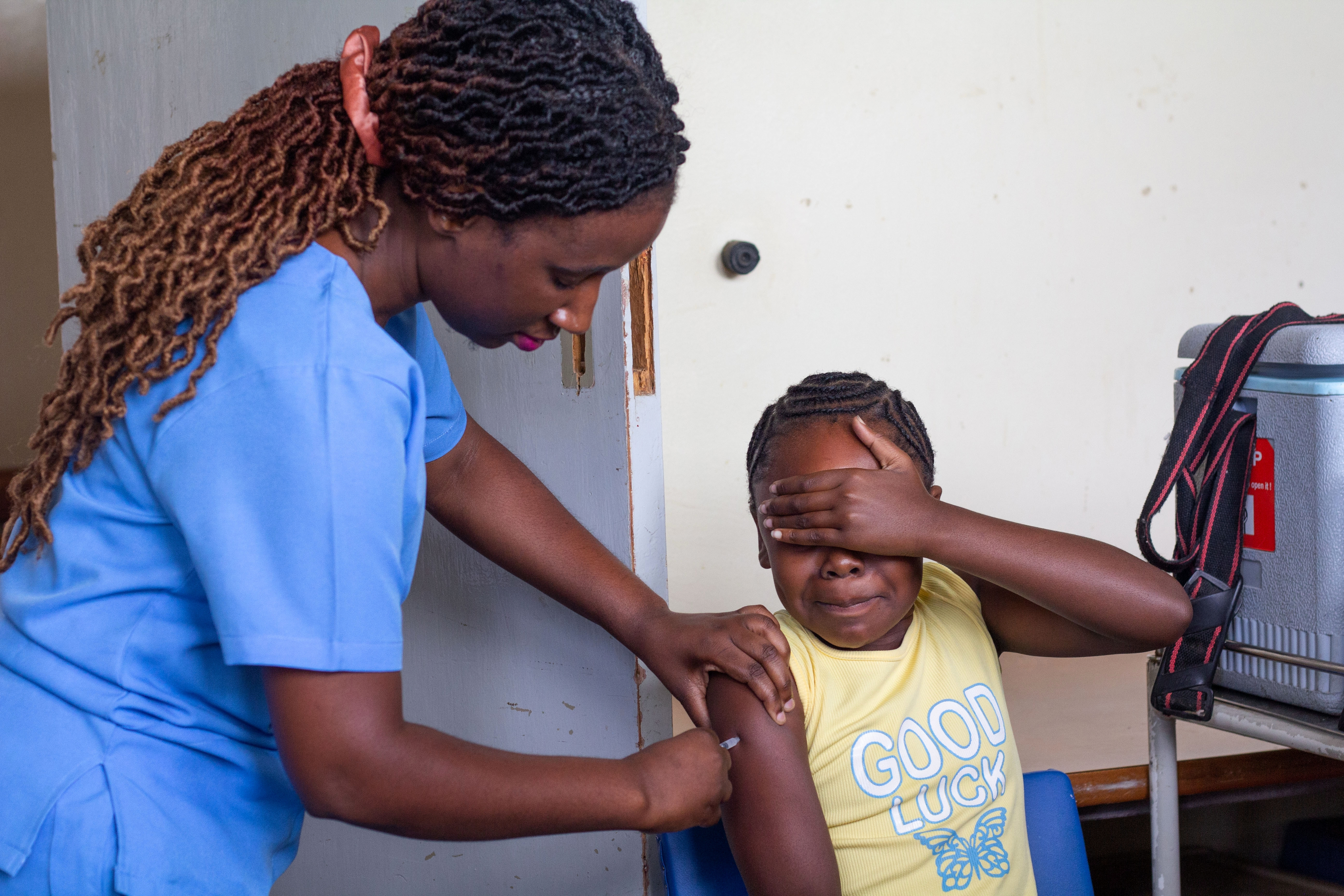
A girls’ soccer tournament in Zimbabwe promotes HPV vaccine awareness, using sport to educate young players and communities about preventing cervical cancer.
![]() Jan 26
Jan 26
by Reid Ashton

FDA launches a new regulatory pathway to approve personalized gene and cell therapies using smaller patient data sets, accelerating access for rare diseases and unmet medical needs.
![]() Nov 12
Nov 12
by Reid Ashton